Hierarchical 3D NiO–CdS heteroarchitecture for efficient visible light photocatalytic hydrogen generation†
Abstract
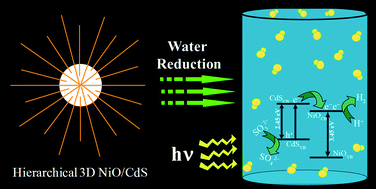
A novel hierarchical three dimensional (3D) NiO–CdS heteroarchitecture was successfully synthesized by a facile single pot hydrothermal route and characterized by

 Please wait while we load your content...
Please wait while we load your content...