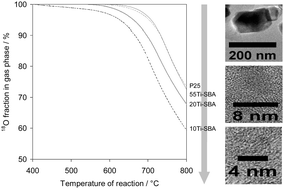
TiO2/SiO2 nanocomposites are synthesized and fully characterized after thermal stabilization at temperatures between 400 °C and 800 °C. The control of the impregnation media is crucial to obtain nanocomposites of satisfactory quality, i.e. presenting no segregation of titania particles outside the silica pore structure. Characterization shows that pore size and pore volume decrease linearly with an increase in titania loading, and remain close to the theoretical values calculated assuming the formation of a non-porous coating. Surface area remains unchanged whatever the titania loading (always comprised between 450 and 480 m2 g−1), and micropore volume evolution suggests the formation of nanometric particles within the silica pores. While X-ray diffraction is inefficient to identify the titania phase, Raman spectroscopy showed the formation of anatase particles, with crystal sizes in the nanometric range (<4.5 nm, when stabilized at 400 °C). Satisfying thermal stability is obtained on the low titania loading nanocomposites (20 wt% TiO2), with only minor anatase crystal growth up to 800 °C. Further characterization by FT-IR of the surface chemical properties of the nanocomposites showed properties similar to that of conventional titania, while improved oxygen mobilities (as evaluated by the 18O/16O exchange reaction) are reported on the low titania loading, thermally stable, composites.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...