Influence of the core structure on the development of polar order and superstructural chirality in liquid crystalline phases formed by silylated bent-core molecules: naphthalene derivatives†‡
Abstract
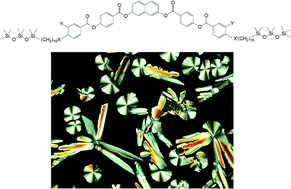
Bent-core molecules based on a bent 2,7-disubstituted naphthalene unit and heptamethyltrisiloxane units at one or both ends of the aliphatic side chains were synthesized and investigated by polarized light microscopy, differential scanning calorimetry, X-ray scattering and electrooptical investigations. The effect of fluorine substitution at the periphery of the rigid core and of the replacement of ether oxygens by carboxylate groups as linking units between the aromatic core and the flexible terminal chains was also investigated. All compounds form broad regions of liquid crystalline phases. Different types of non-polar and polar, i.e. antiferroelectric and ferroelectric switching smectic phases, and columnar phases were observed, depending on the molecular structure. Most interestingly, a temperature dependent transition from an antiferroelectric switching to a ferroelectric switching oblique columnar phase was observed. All double-silylated compounds show switching by a collective rotation around the molecular long axis, a process which switches the superstructural chirality in these LC systems.

 Please wait while we load your content...
Please wait while we load your content...