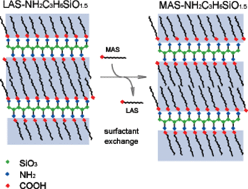
A new layered organosilicate with a multi-layer LB film-like structure, in which the carboxyl groups are connected to each other via a 3-aminopropylsilicate layer, is successfully synthesized by a self-assembly process using alkanoic acid (CnH2n+1COOH, n > 10) combined with the co-condensation of 3-aminopropyltriethoxysilane. The structure is elucidated by elemental analysis, X-ray diffraction, 13C and 29Si MAS-NMR, infrared spectroscopy and thermogravimetry. The combined results of these techniques demonstrate that the alkyl groups are not packed in a crystalline state, but exist in a liquid-like state in a well-developed lamellar structure with the formula CnH2n+1COOHNH2CH2CH2CH2SiO1.5. In the extremely thin siloxane layer that is developed in the lamellar structure, nearly all of the silicon atoms are bound to one carbon atom, three -OSi groups and no -OH groups. This structure (one silicon atom bound to one 3-aminopropyl group) possibly contains the most highly-populated aminopropyl group among all of the aminopropylsilicates that have ever been reported. The LB film-like interlayer can be exchanged with other alkanoic acids to form another lamellar structure in the same series that possesses the appropriate layer spacing. A fast delamination (<5 min) process is observed after the addition of Fe3+, which implies a rapid diffusion of transition metal cations, and which is also consistent with the liquid-like nature of the alkyl group assembly. The specific amount of Fe3+ sorption reaches 5.1 mmol g−1, probably the highest adsorption capacity ever reported among the aminopropylsilicates.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...