Hydrolysis study of fluoroorganic and cyano-based ionic liquid anions – consequences for operational safety and environmental stability
Abstract
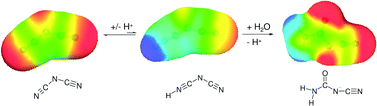
The hydrolytic stability of ionic liquid anions is a key property with regard to their technical applicability and environmental stability. From a technical point of view hydrolytic processes may lead to reduced durability, diminished technical performance and reduced operational safety in that corrosive and/or toxic hydrolysis products are formed. On the other hand, susceptibility to hydrolytic processes is advantageous where environmental stability and persistency are concerned, since hydrolysis is the most important abiotic degradation pathway in the environment. We investigated the hydrolytic stability of the most common ionic liquid anions, dicyanimide [N(CN)2]−, tricyanmethanide [C(CN)3]−, tetracyanidoboranate [B(CN)4]−, bis(trifluoromethylsulphonyl)imide [(CF3SO2)2N]−, trifluorotris(pentafluoroethyl)phosphate [(C2F5)3PF3]− and 1,1,2,2-tetrafluoroethanesulphonic acid [H(C2F4)SO3]−, as a function of pH (1, 7, 9 and 13) and temperature. The results show that there was no difference in hydrolytic stability as recorded for 1-ethyl-3-methylimidazolium (IM12) or for the alkali cations. All the anions were stable under neutral and slightly basic conditions (half-lives at 25 °C ⇒1 year). In strongly acidic and basic solutions, however, B(CN)4−, (CF3SO2)2N−, (C2F5)3PF3− and H(C2F4)SO3− were hydrolytically stable, whereas N(CN)2− and C(CN)3− were not. The kinetics of hydrolysis were recorded and Arrhenius plots were generated for the latter two anions. In addition, their hydrolysis pathways and the resulting products were identified via mass spectrometry. The cytotoxicity of hydrolysed IL solutions towards the mammalian cell line IPC-81 and the identified hydrolysis products (pure compounds) was investigated for a first estimate of their toxicological properties.

 Please wait while we load your content...
Please wait while we load your content...