Nitrile hydratase CLEAs: The immobilization and stabilization of an industrially important enzyme†
Abstract
The successful immobilization and stabilization of a
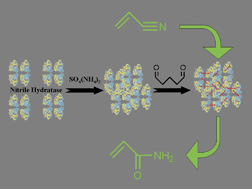
- This article is part of the themed collection: Green and sustainable chemistry

 Please wait while we load your content...
Please wait while we load your content...