Detoxification of aryl-organochlorine compounds by catalytic reduction in supercritical carbon dioxide
Abstract
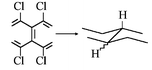
Pentachlorophenol, octachloronaphthalene and decachlorobiphenyl were smoothly converted to cyclohexanol, decalin and dicyclohexyl, respectively, by reaction, during 0.5–2 h, with excess hydrogen over alumina-supported palladium (5% w/w) in the presence/absence of supercritical carbon dioxide (50–90 °C). With these conditions, dechlorinations and dearomatization to their cyclic analogs were complete but no carbon–carbon bond scission was observed and only traces of partial deoxygenation/dimerization of the pentachlorophenol substrate (to form 1,1′-oxybiscyclohexane) was seen. The scCO2 medium functioned as an inert support for the reactions; differences in rates of reaction between chlorinated compounds and their aromatic hydrocarbon homologs were not observed. These results suggest that environmentally recalcitrant chlorinated aromatic compounds can be detoxified facilely by catalytic reduction with H2 under mild conditions.

 Please wait while we load your content...
Please wait while we load your content...