Influence of biochar thermal regeneration on sulfamethoxazole and dissolved organic matter adsorption†
Abstract
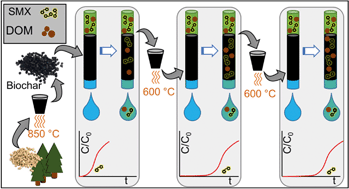
Biochar is emerging as a cost-effective, environmentally-sustainable adsorbent for removing organic contaminants (OC) from wastewater, stormwater, and drinking water, but strategies for managing exhausted biochar are needed. Here, pine biochar generated at 850 °C was exhausted by background dissolved organic matter isolated from surface water [dissolved organic carbon ∼4.2 mg L−1, UV-absorbance at 254 nm (UVA254) ∼0.10 cm−1] and sulfamethoxazole (SMX) [∼200 ng L−1], in a column. Exhausted biochar underwent a semi-oxic-thermal-regeneration step at 600 °C (i.e., heat treatment). SMX and UVA254 adsorption capacity and breakthrough were evaluated in rapid small-scale column tests (RSSCTs). Relative to fresh biochar, heat treated biochar that had been exhausted exhibited ∼3.5-fold and ∼3-fold greater SMX and UVA254 adsorption capacities, respectively, and ∼3-fold increase in adsorption efficiency (i.e., mass loss-adjusted SMX adsorption capacity). When applying the heat treatment to fresh biochar, a similar improvement in adsorption capacity was observed. Adsorption capacity and BET surface area were positively correlated and continued to increase after a second exhaustion–regeneration cycle, but the adsorption efficiency remained the same due to mass loss. SMX breakthrough correlated with that of UVA254, which provides the basis for a rapid, inexpensive method to predict OC breakthrough. Heat-treated biochar's SMX adsorption capacity was ∼20% of activated carbon's. Greater empty-bed-contact times increased the SMX adsorption capacity of heat-treated biochar. These results suggest that thermal regeneration could enhance the economic and environmental sustainability of biochar sorbents.


 Please wait while we load your content...
Please wait while we load your content...