Inorganic–organic layer by layer hybrid membranes for lithium–sulfur batteries†
Abstract
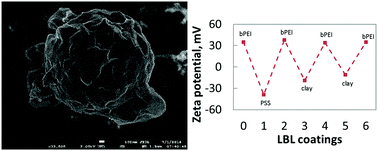
The sulfur cathode has a gravimetric theoretical energy of 3500 W h kg−1 which is 3.5× higher than that of the most energy dense cathode material used in commercial lithium ion batteries (1000 W h kg−1 for LiNi0.8Co0.15Al0.05O2 (NCA)). The lithium–sulfur battery also promises a dramatic drop in the cost of high energy density batteries due to the wide availability of sulfur which may compensate costs associated with the operation of lithium metal sheets or powder anodes which are not stable in moist air and require dry rooms or an inert atmosphere. High sulfur loading cathodes have been typically assembled by infiltrating sulfur into porous conductive hosts or interlayers. However, a different approach is that of synthesizing sulfur particles in situ by the reaction of strong acids and sulfur precursors such as sodium thiosulfate. In this communication we propose (A) a novel, eco-safe method for the bottom-up synthesis of sulfur particles which is based on the reaction of weak acids such as vitamin C and commercial potassium polysulfides; (B) layer by layer assembly of hybrid inorganic/organic membrane encapsulating the sulfur particle impedes polysulfide dissolution or protecting the lithium metal anode; (C) a close relationship between the utilization of the lithium metal anode and the short cycle life of lithium–sulfur cells.


 Please wait while we load your content...
Please wait while we load your content...