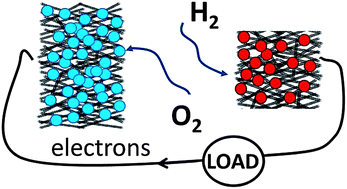
The unusual ability of O2-tolerant hydrogenases (H2ase) to produce electricity from a H2–air mixture (when used as the anodic electrocatalyst in a simple, membrane-less fuel cell) is investigated with the aim of establishing a strategy for raising volume power density, the measure of importance for miniature devices. Compacted mesoporous carbon electrodes provide a simple and inexpensive method for obtaining a large increase in productive enzyme loading, greatly increasing current densities and stability. Operated under a 78% H2–22% air mixture at 25 °C, typical current densities at a stationary H2ase anode and bilirubin oxidase cathode are 4.60 ± 0.32 mA cm−2 and 1.23 ± 0.12 mA cm−2, respectively. The power limitation due to low O2 concentration is addressed by re-proportioning the cathode/anode area ratio to balance the cathodic and anodic currents. At room temperature, the maximum power density of the fuel cell with an anode/cathode (A/C) ratio of 1 : 3 (1A/3C) is 1.67 ± 0.24 mW cm−2 (per anode area) or 0.42 ± 0.06 mW cm−2 (per total area). Good prospects for stability are demonstrated by the fact that 90% of the power is retained after continuously working for 24 h, and more than half of the power is retained after one week of non-stop operation. Using an even weaker O2 mixture (89% H2, 11% air) the 1A/3C cell gives over 0.8 mW cm−2 (anode) or 0.2 mW cm−2 (total electrode area). The results demonstrate the feasibility of membrane-less hydrogen–air fuel cells delivering volume power densities well in excess of 1 mW cm−3.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...