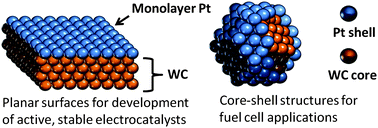
The high cost and limited supply of platinum (Pt) represent barriers to large-scale commercial applications of several electrochemical devices that utilize Pt as electrocatalysts. In this Perspective we discuss opportunities to replace all but the top atomic layer, or monolayer (ML), of Pt with low-cost, “Pt-like” tungsten carbide (WC). These ML Pt-WC catalysts represent the lower limit of Pt loading, allowing for a significant decrease in Pt costs in devices such as fuel cells and electrolyzers. Density functional theory (DFT) calculations and experimental measurements indicate that the ML Pt-WC surface exhibits chemical and electronic properties that are very similar to bulk Pt for simple reactions such as the hydrogen evolution reaction, although deviation in behavior is observed for more complex reactions such as the electrooxidation of methanol. In order to further improve the performance of ML Pt-WC electrocatalysts and transfer knowledge gained from thin films to high surface area catalysts, a better understanding of the electronic, electrochemical, and catalytic properties of ML Pt-WC is needed. This Perspective provides an overview of these promising ML Pt-WC electrocatalysts and discusses approaches to overcoming limitations based on a better understanding of the Pt-WC interface and its behavior in electrochemical environments.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...