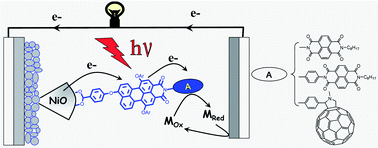
We report on the synthesis, electrochemical, photophysical, and photovoltaic properties of a series of three organic dyads comprising a perylene monoimide (PMI) dye connected to a naphthalene diimide (NDI) or a fullerene (C60) for application in dye-sensitized solar cells (DSCs) with nanocrystalline NiO electrodes. It was found that the secondary electron acceptor (NDI or C60) in all the three dyads extends the charge separated state lifetime by about five orders of magnitude compared to the respective parent PMI dye. Nanosecond pump–probe experiments of the NiO/dyads in the presence of the electrolyte show that the reduction of triiodide by the secondary electron acceptor is slow in all the dyads, which we ascribe to a weak driving force for this reaction. This reaction is significantly faster with the cobalt electrolyte (tris(4,4′-di-tert-butyl-2,2′-bipyridine)cobalt(II/III)), whose driving force is larger; however, its reaction with the reduced dyads is still rather slow. We demonstrate that the larger photovoltage observed with the cobalt electrolyte (VOC = 285 mV) relative to the iodide electrolyte (VOC = 120 mV) is due to a decrease in the dark current for the former owing to slower interfacial electron transfer of the reduced mediator with the injected holes into the NiO electrode. In terms of photovoltaic performances, the most efficient dyad is the system in which the NDI is directly connected to the PMI (η = 0.14% under AM 1.5 with the cobalt electrolyte), but the dyad containing the fullerene acceptor exhibits the highest IPCE and the highest short circuit current density (IPCE = 57%, JSC = 1.88 mA cm−2) with the iodide electrolyte. The latter performances are attributed to the slightly stronger reducing power of C60 relative to NDI, which favours the reduction of the mediator in the electrolyte.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...