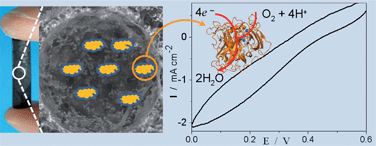
In the present study we have shown that carbonaceous micro/macrocellular foams can be used for efficient and stable non-specific enzyme entrapment. In this context, Bilirubin Oxidase adsorbed into the porous electrode is able to reduce O2 to water and electrons are transferred directly from the electrode to the enzyme without the need of a redox mediator. The reduction current is stable for several days under continuous operation and therefore we consider the carbonaceous foams are very promising candidates for the construction of 3-dimensional biofuel cell cathodes. Mediator free, the electrode preparation and further enzyme adsorption are extremely simple, low cost and versatile. Most important, the excellent mechanical strength and the synthetic route allow us to design at will the size and external shape of the electrodes, which are of vital importance if we wish to incorporate electrodes into devices. The Integrative Chemistry synthetic route allows accessing hierarchical porosities with the advantage of specific functionalities. Macropores serve to fuel transport and micropores as anchoring sites for enzyme entrapment. Despite the inability to reach the Levich diffusion limited current suggesting that the porosity is not yet fully optimized, the results presented here show that porous carbonaceous foam electrodes allow for an incredible increase in enzyme loading which allows for a 500-fold current enhancement and stabilization of the direct electron transfer current from few hours to several days as compared to conventional flat electrodes.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...