Unprecedented tetranuclear complexes with “weighing balance shaped” topology: single crystal structures, unusual EPR spectra, magnetic properties and antioxidant activity†
Abstract
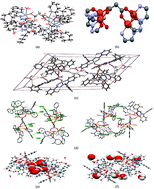
Using the 2-hydroxy-N′-[(E)-phenyl(pyridin-2-yl)methylidene]benzohydrazide ligand and dicarboxylate anions (succinate and malonate), two new copper(II) complexes of formula [Cu4(μ-L)4(μ-CH2(COO)2)(H2O)3](NO3)2·4H2O (1) and [Cu4(μ-L)4(μ-CH2–CH2(COO)2)(H2O)](NO3)2] (2) have been synthesized and characterized by single crystal X-ray diffraction. Complexes 1 and 2 contain a dicarboxylate bridged tetranuclear Cu(II) cationic unit with “weighing balance shaped” topology. These compounds have also been studied by variable temperature magnetic susceptibility measurements, X-band EPR spectroscopy and electrochemistry. The unprecedented tetranuclear copper complexes 1 and 2 exhibit antiferromagnetic interactions between adjacent copper(II) centers with magnetic exchange coupling constants (J) of J1 = −6.8 cm−1, J2 = −20.6 cm−1 and g = 2.03 for 1 and J1 = −3.1 cm−1, J2 = −13.9 cm−1 and g = 2.02 for 2. The goal for our research work is to understand the factors that govern the stability of the Cu–O(–O–C–O–) bond towards reactive oxygen species (ROS) in Cu-containing mimics utilizing a systematic approach. The antioxidant superoxide dismutase measurements indicate that these complexes behave as superoxide dismutase mimics.


 Please wait while we load your content...
Please wait while we load your content...