Temperature-induced 1D lanthanide polymeric frameworks based on Lnn (n = 2, 2, 4, 6) cores: synthesis, crystal structures and luminescence properties†
Abstract
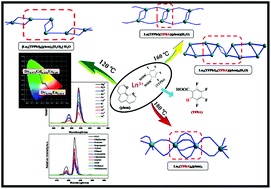
The hydrothermal reaction of the same reactive system containing Ln(NO3)3·6H2O, tetrafluorophthalic acid (H2TFPht), and 1,10-phenanthroline (phen) at different temperatures yielded coordination polymers, [Ln2(TFPht)3(phen)2(H2O)2]·H2O (Ln = Sm 1, Eu 2, Gd 3, Tb 4, Dy 5; at 120 °C), La(TFPht)(TFBA)(phen)(H2O) (6; at 160 °C), Ln3(TFPht)4(TFBA)(phen)3(H2O)3 (Ln = Dy 7, Tb 8, Yb 9; at 160 °C), and Ln2(TFBA)6(phen)2 (Ln = Eu 10, Tb 11; at 180 °C). 2,3,4,5-Tetrafluorobenzoic acid (TFBA) was produced from the decarboxylation of TFPht. X-ray structural analysis reveals that these compounds contain different structural motifs. Complexes 1–5 exhibit zigzag chain structures based on the center-related tetranuclear [Ln4] as SBUs (Secondary Building Units) with two crystallographically independent Ln3+ ions. Complex 6 contains a double chain structure with center-related binuclear [La2] as SBUs. Complexes 7–9 show single chain structures involving center-related hexanuclear [Ln6] as SBUs with three crystallographically independent Ln3+ ions. Complexes 10 and 11 have ribbon chain structures involving binuclear [Ln2] as SBUs with two crystallographically independent Ln3+ ions. The photoluminescence properties of complexes 1–5 were studied. The Eu3+ and Tb3+ complexes exhibit bright red and green emissions with quantum yields of 15.87% for 2 and 23.82% for 4. The two-component Dy:Eu- and three-component Gd:Dy,Eu-doped complexes provided white light emission. Moreover, 2 could be a potential luminescent probe for detecting nitrobenzene and Ni2+ ion through significant fluorescence decrease of Eu3+.

 Please wait while we load your content...
Please wait while we load your content...