pH dependent facile synthesis of di- and trinuclear oxime based Cu(ii) complexes: antiferromagnetic coupling in the dinuclear cores and spin frustration in the triangular core†
Abstract
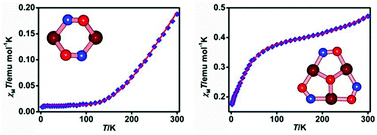
An oxime based tridentate Schiff base ligand 3-[3-(dimethylamino)propylimino]butan-2-one oxime (HL) produced two dinuclear compounds [Cu2L2(H2O)](ClO4)2 (1) and [Cu2L2(H2O)](BF4)2 (2), and a hexanuclear compound [{Cu3(HL)3(O3ClO)(μ3-O)}2(μ-H)](ClO4)7 (3) when it was reacted with Cu(ClO4)2·6H2O or Cu(BF4)2·6H2O at different pH values. All three compounds have been structurally and magnetically characterized. Compounds 1 and 2 are dinuclear species in which the two square planar copper(II) ions are joined solely by the double oximato bridges. On the other hand, the hexanuclear compound 3 consists of two triangular Cu3O cores held together by a proton separated by an O⋯O distance of 2.498(10) Å. The three square pyramidal copper(II) ions at the corners of an isosceles triangle form a triangular core through central oxido (μ3-O) and peripheral oximato bridges. Each triangular Cu3O core is capped by an unusual triply coordinated (μ3-perchlorato-O,O′,O′′) perchlorate anion. Variable-temperature (2–300 K) magnetic susceptibility measurements show that compounds 1–3 exhibit a strong antiferromagnetic interaction with J values −562.6, −633.1 and −636.0 cm−1 respectively. The X-band EPR data at low temperature clearly indicate the presence of a spin frustration phenomenon in complex 3.

 Please wait while we load your content...
Please wait while we load your content...