High-throughput ultrasonic synthesis and in situ crystallisation investigation of metal phosphonocarboxylates†
Abstract
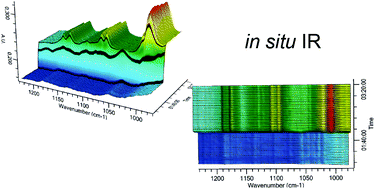
High-throughput reactions of aqueous solutions of 4-(phosphonomethyl)benzoic acid (H3PMBC) with metal salts were carried out using, for the first time, ultrasonication as well as the previously described conventional heating. On screening the influence of 12 divalent metal ions, two new compounds ([Ca(H2O)2(H2PMBC)2] and [Ni(H2O)4(H2PMBC)2]) were discovered. Their formation fields were established by systematically studying the systems Ni2+ or Ca2+/H3PMBC/KOH. Both compounds crystallise in the space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ([Ca(H2O)2(H2PMBC)2]: a = 4.6806(9) Å, b = 6.1340(12) Å, c = 17.932(4) Å, α = 83.84(3)°, β = 89.03(3)°, γ = 85.65(3)°; [Ni(H2O)4(H2PMBC)2]: a = 4.7713(10) Å, b = 6.8621(14) Å, c = 16.598(3) Å, α = 83.42(3)°, β = 89.71(3)°, γ = 83.50(3)°) and contain MO6-octahedra linked to form chains by hydrogen bonded (H2PMBC−)2-dimers formed between the carboxylate groups. The formation of [Ni(H2O)4(H2PMBC)2] was studied by in situ EDXRD and in situ IR spectroscopy. The increased formation rate of [Ni(H2O)4(H2PMBC)2] under ultrasonic irradiation as compared to conventional heating could be unequivocally attributed to the faster dissolution of the ligand H3PMBC.
([Ca(H2O)2(H2PMBC)2]: a = 4.6806(9) Å, b = 6.1340(12) Å, c = 17.932(4) Å, α = 83.84(3)°, β = 89.03(3)°, γ = 85.65(3)°; [Ni(H2O)4(H2PMBC)2]: a = 4.7713(10) Å, b = 6.8621(14) Å, c = 16.598(3) Å, α = 83.42(3)°, β = 89.71(3)°, γ = 83.50(3)°) and contain MO6-octahedra linked to form chains by hydrogen bonded (H2PMBC−)2-dimers formed between the carboxylate groups. The formation of [Ni(H2O)4(H2PMBC)2] was studied by in situ EDXRD and in situ IR spectroscopy. The increased formation rate of [Ni(H2O)4(H2PMBC)2] under ultrasonic irradiation as compared to conventional heating could be unequivocally attributed to the faster dissolution of the ligand H3PMBC.

 Please wait while we load your content...
Please wait while we load your content...