Dinuclear gold(i) dithio- and diselenophosph(in)ate complexes forming mononuclear gold(iii) oxidative addition complexes and reversible chemical reductive elimination products†
Abstract
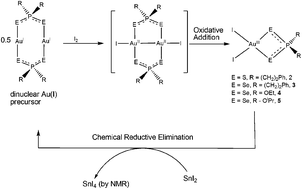
Dinuclear gold(I) dithio- and diselenophosph(in)ate complexes were prepared to serve as precursors for subsequent oxidative addition (OA) chemistry following reaction with mild oxidant iodine, I2. The new OA products circumvented the formation of the expected dinuclear Au(II) complexes, but instead formed novel chelating mononuclear square-planar gold(III) products of the type [AuI2{E2PR2}] (R = (CH2)2Ph; E = S, 2; E = Se, 3) and [AuI2{Se2P(OR)2}] (R = Et, 4; iPr, 5) directly. We further demonstrate that this process is chemically reversible as all the Au(III) complexes undergo chemical reductive elimination to the starting dinuclear Au(I) complexes in the presence of SnI2 as determined by 119Sn and 31P NMR. The complexes (2–5) were all prepared from the reaction between I2 and the corresponding dinuclear gold(I) precursor material. All new complexes were characterized by 1H, 31P, 77Se (for E = Se) NMR, infrared spectroscopy, elemental analysis and single crystal X-ray diffraction.

 Please wait while we load your content...
Please wait while we load your content...