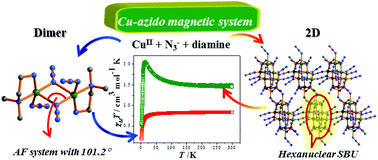
This article describes syntheses, structural characterizations and magnetic studies of two different Cu(II)–azido compounds, a discrete dinuclear complex and an extended 2D network. The compounds, [Cu(μ1,1-N3)(N3)(Me2en)]2 (1) and [Cu3(μ1,1,1-N3)2(μ1,1,3-N3)(μ1,1-N3)2(μ1,3-N3)(Me2en)]n (2), have been synthesized by controlling the relative concentration of the blocking ligand, N,N-dimethylethylenediamine (Me2en). Compound 1 is a dinuclear compound which is formed by a doubly asymmetric μ1,1-N3 bridging ligand, while 2 is a rare Cu–azido system where four different types of binding modes of azide ligands are present in a single compound. Compound 2 contains a hexanuclear core, where the Cu(II) centres are connected to each other by μ1,1,1, μ1,1 and μ1,1,3 bridging azide ligands. The hexanuclear core acts as a secondary building block and further assembles via μ1,3 and μ1,1,3 azide groups, forming a 2D network in the crystallographic ac plane. Interestingly, temperature-dependent magnetic study suggests that the dinuclear compound 1 exhibits an antiferromagnetic interaction through the μ1,1-N3 bridge, which has also been supported by density functional theory (DFT) calculations. In the case of 2, an overall dominant ferromagnetic interaction is observed while antiferromagnetic interaction operates between the hexanuclear cores.

 Please wait while we load your content...
Please wait while we load your content...