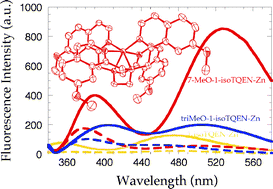
Previously, we have reported that 1- and 3-isoTQENs (N,N,N′,N′-tetrakis(1- or 3-isoquinolylmethyl)ethylenediamines) exhibit a specific fluorescence enhancement toward zinc ion. In this study, three methoxy-substituted derivatives of 1-isoTQEN were synthesized and their fluorescent response toward zinc ion was studied. The substitution pattern of the methoxy group significantly changes the solubility of compounds in aqueous DMF, λmax in the absorption spectra, excitation/emission wavelengths and fluorescence intensity of zinc complexes. In the presence of zinc ion, 7-MeO-1-isoTQEN exhibits higher fluorescence intensity and longer excitation/emission wavelengths (λex = 342 nm, λem = 526 nm) than 6-MeO-1-isoTQEN (λex = 303 nm, λem = 469 nm) and 5,6,7-triMeO-1-isoTQEN (λex = 340 nm, λem = 504 nm). The fluorescence intensity of a zinc complex of 7-MeO-1-isoTQEN (ϕ = 0.122) is four times higher than the parent 1-isoTQEN (ϕ = 0.034) under the same conditions. The crystal structure of 7-MeO-1-isoTQEN-Zn complex reveals that all six nitrogen atoms participate to the metal coordination with ideal octahedral geometry, affording significantly high metal binding affinity comparable with TPEN (N,N,N′,N′-tetrakis(2-pyridylmethyl)ethylenediamine). 7-MeO-1-isoTQEN detects zinc ion concentration change in cells by fluorescence microscopic analysis.

 Please wait while we load your content...
Please wait while we load your content...