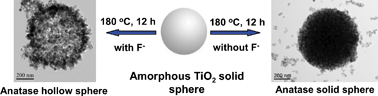
Mesporous anatase-phase TiO2 hollow spheres with high photocatalytic activity were prepared by hydrothermal treatment and self-transformation of amorphous TiO2 solid spheres in an NH4F aqueous solution. The prepared samples were characterized by X-ray diffraction, scanning and transmission electron microscopy, X-ray photoelectron spectroscopy, N2 adsorption-desorption isotherms and UV-vis absorption spectroscopy. The photocatalytic activity was evaluated by photocatalytic oxidation decomposition of acetone in air under UV irradiation. It is found that F− plays an essential role in the formation of TiO2 hollow spheres. F− not only induces the hollowing of TiO2 solid spheres, but also promotes the crystallization of anatase TiO2 nanocrystals. A possible formation mechanism for the TiO2 hollow spheres by localized Ostwald ripening or chemically induced self-transformation is proposed based on the experimental observations. Furthermore, the molar ratios of NH4F to TiO2 (R) exhibit a significant influence on the morphology and photocatalytic activity of the TiO2 samples. The photocatalytic activity of the samples prepared in the presence of NH4F is higher than that of TiO2 sample prepared in pure water and commercial Degussa P25 (P25) powders. Especially, the TiO2 hollow spheres prepared at R = 1 show the highest photocatalytic activity and the specific rate constant Sk exceeds that of P25 by a factor of more than two.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...