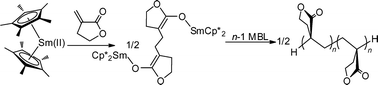
This contribution reports the first study of coordination-addition polymerization of renewable butyrolactone-based vinyl monomers, MBL (α-methylene-γ-butyrolactone) and MMBL (γ-methyl-α-methylene-γ-butyrolactone), using neutral lanthanocene(II), non-lanthanocene(III), and cationic group 4 metallocene catalysts. The samarocene(II) catalyst, Cp*2Sm(THF)2, promotes a rapid, efficient, and controlled polymerization of MBL and MMBL in DMF at ambient temperature, exhibiting a high TOF of 3000 h−1, typically near quantitative initiator efficiency, and the ability to control the polymer MW. The resulting atactic PMBL and PMMBL have high Tg's of 194 °C and 227 °C, respectively; when compared to atactic PMMA having comparable MW, the Tg and onset decomposition temperatures of the PMMBL produced are substantially higher (by ∼120 °C and 40 °C, respectively). Owing to the living/controlled characteristics of this polymerization, well-defined random and block copolymers of MBL with MMA and MMBL can be readily synthesized. Results of the kinetic and polymerization studies indicate that the true active species is the trivalent samarocene centers attached to the single growing polymer chain, derived presumably from a redox-then-radical-coupling process. In comparison, the polymerizations by non-lanthanocene(III) silylamides, Ln[N(SiMe3)2]3 (Ln = La, Nd, Sm, Er), and by cationic group 4 metallocene and half-metallocene catalysts incorporating C2 and Cs symmetric ligands are much slower and less effective. Catalytic polymerization of MBL by Cp*2Sm(THF)2 has also been realized in the presence of an enolizable organo acid as a suitable chain transfer agent.

 Please wait while we load your content...
Please wait while we load your content...