Synthesis, structures and ring-opening polymerization studies of new zinc chloride and amide complexes supported by amidinate heteroscorpionate ligands†
Abstract
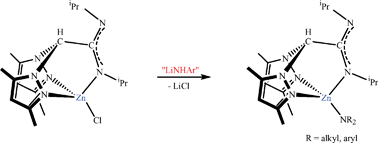
The reaction of the heteroscorpionate lithium salts [Li(pbpamd)(THF)] [pbpamd = N,N′-diisopropylbis(3,5-dimethylpyrazol-1-yl)acetamidinate] and [Li(tbpamd)(THF)] [tbpamd = N-ethyl-N′-tert-butylbis(3,5-dimethylpyrazol-1-yl)acetamidinate] with 1 equivalent of ZnCl2 in THF affords very high yields of the neutral heteroscorpionate chloride zinc complexes [ZnCl(NNN)] (NNN = pbpamd 1 and tbpamd 2). Compound 1 was used as a convenient starting material for the synthesis of aromatic amide zinc compounds [Zn(NHAr)(pbpamd)], where NHAr = 4-methylphenylamide (NH-4-MeC6H4) 3, 2,4,6-trimethylphenylamide (NH-2,4,6-Me3C6H2) 4 and 2,6-diethylphenylamide (NH-2,6-Et2C6H3) 5, by the reaction of the corresponding aromatic primary amide lithium salts. Alternatively, aliphatic amide derivatives [Zn(NR2)(pbpamd)] (R = SiMe36, SiHMe27 and iPr 8) were cleanly prepared by reacting the amidine-heteroscorpionate compound Hpbpamd with the corresponding bis(amide) zinc complexes [Zn(NR2)2] (R = SiMe3, SiHMe2 and iPr). The single-crystal X-ray structures of complexes 2, 3 and 6 confirm a 4-coordinate arrangement in all cases, with the zinc metal surrounded in a distorted tetrahedral geometry and the heteroscorpionate ligands arranged in a κ3 coordination mode. Whereas aliphatic amide heteroscorpionates 6–8 can act as efficient single-component initiators for the ring-opening polymerization of ε-caprolactone at room temperature, aromatic amide derivatives were not capable of yielding polymers even at high temperature. ε-Caprolactone is polymerized within minutes to give medium–high molecular weight polymers under mild conditions and with narrow polydispersities (Mw/Mn = 1.26). Polymer end group analysis shows that the polymerization mediated by aliphatic amide zinc complexes is initiated by amide transfer to the monomer.

 Please wait while we load your content...
Please wait while we load your content...