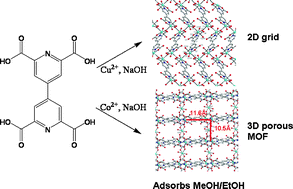
4,4′-Bipyridine-2,6,2′,6′-tetracarboxylic acid H4L·3H2O, (1) and its copper(II) and cobalt (II) coordination polymers [Cu2L(H2O)4]2n (2) and {[Co(H2O)6]·[Co3L2(H2O)2]·10H2O}3n (3) have been hydrothermally synthesized. Compound 1 packs into a crystal viaH-bonds. Complex 2 is a 2D coordination grid, in which Cu is in an elongated octahedral constructed by a mer-geometry pyridyl-2,6-dicarboxylate, one bridging carboxylate oxygen and two water molecules. The L4− is central symmetrical. Each pyridyl-2,6-dicarboxylate chelete to Cu(II) and one carboxylate bridge Cu(II) ions in a 1,1-fashion. The 2D coordination polymer links to adjacent layers viaH-bonds. Complex 3 is a metal–organic framework with 11.6 × 10.5 Å rectangular channels. All Co(II) ions are located in an octahedral coordination environment. The rectangular channels are composed of Co2+-L4− walls and the walls are linked through carboxylate oxygen atoms and Co(II) ions. Co(H2O)62+ as counter ions fill in porous channel. The removal of water molecules from 3 at 210 °C or higher affords the porous material [Co4L2]n, which can adsorb 16 (36%) methanol or 9 (32%) ethanol in the vapour phase. The porosity of 3 is higher than reported 4,4′-bipyridine-2,6,2′,6′- tetracarboxylate complexes. The framework of 3 remains but the crystallinity is lost upon removal of the H2O molecules. The dehydrated framework of 3 partially recovery crystallinity after it adsorbs H2O, CH3OH and C2H5OH. The methanol in the framework can be replaced by H2O reversibly.

 Please wait while we load your content...
Please wait while we load your content...