Electronic coupling in 1,4-(COS)2C6H4 linked MM quadruple bonds (M = Mo, W): the influence of S for O substitution†
Abstract
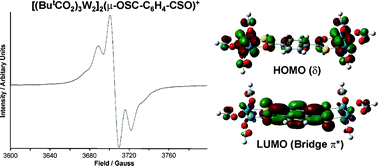
Electronic structure calculations employing density functional theory on the compounds [(HCO2)3M2]2(µ-X–C6H4–X) where M = Mo and W and –X = –CO2, –COS and –CS2 reveal that the successive substitution of oxygen by sulfur leads to enhanced electronic coupling as evidenced by the increased energy separation of the metal δ orbital combinations which comprise the HOMO and HOMO−1. This enhanced coupling arises principally from a lowering of the LUMO of the X–C6H4–X bridge which, in turn, increases

 Please wait while we load your content...
Please wait while we load your content...