Inhibition of Thermus thermophilus HB8 thioredoxin activity by platinum(ii)†
Abstract
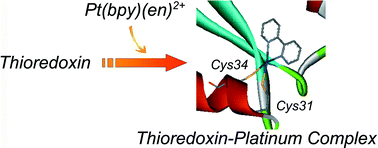
A 1 ∶ 1 thioredoxin–Pt(bpy) complex 1 was prepared by adding [Pt(bpy)(en)]Cl2 (bpy = 2,2′-bipyridine, en = ethylenediamine) to Thermus thermophilus HB8 thioredoxin in pH 8 phosphate buffer. Matrix-assisted laser desorption-ionization time of flight mass spectrometry (MALDI-TOF MS) and UV spectra of 1 indicate the formation of Pt(bpy)(cys-Ala-Pro-cys-containing peptide fragment). These findings suggest that the Pt(bpy)2+ unit binds to the active site of thioredoxin. The thioredoxin–platinum complex has no catalytic activity for the reduction of glutathione disulfide in the presence of NADPH and thioredoxin reductase, so that the platinum complex functions as an inhibitor.

 Please wait while we load your content...
Please wait while we load your content...