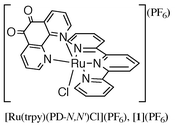
The synthesis and electrochemical properties of a Ru(II) complex having a redox active ligand, 1,10-phenanthroline-5,6-dione, [Ru(trpy)(PD-N,N′)Cl](PF6)
([1](PF6)), and mixed-metal complexes [(PPh3)2Pd(O,O′-PD-N,N′)Ru(trpy)Cl](PF6)
([2](PF6)) and [(PPh3)2Pt(O,O′-PD-N,N′)Ru(trpy)Cl](PF6)
([3](PF6))
(trpy = 2,2′:6′,2″-terpyridine, PD = 1,10-phenanthroline-5,6-dione) are presented. The complex of [1]+, which was prepared by the reaction of Ru(trpy)(DMSO)Cl2 with PD in hot ethanol, underwent two reversible reductions in the PD-based redox reactions. The quinoid moiety of [1]+ was endowed with coordination ability to metals by one- and two-electron reduction of the complex. The mixed-metal complexes of [2]+ and [3]+ were synthesized by the reactions of [1]+ with M(PPh3)4
(M = Pd, Pt), and both complexes have the [1]− and M(II) frameworks.

 Please wait while we load your content...
Please wait while we load your content...