Methane decomposition to tip and base grown carbon nanotubes and COx-free H2 over mono- and bimetallic 3d transition metal catalysts†
Abstract
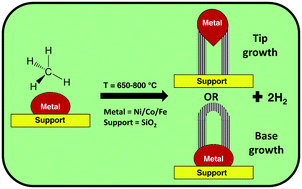
Mono- and bimetallic 3d transition metals (Ni, Fe, Co) over a SiO2 support have been investigated for methane decomposition to COx-free H2 and value added carbon (CNT) in a fixed bed reactor. In our work we synthesized mono/bimetallic Ni, Fe and Co catalysts with various mole ratios. The catalysts were tested under the same reaction conditions for methane decomposition and the influence of their properties on the CNT features was investigated. The synthesized bimetallic catalysts were found to have higher activity and stability than monometallic catalysts. Among the bimetallic catalysts, those with higher Ni content (9Ni–1Fe/SiO2 and 9Ni–1Co/SiO2) exhibited higher methane conversion. The properties of fresh and spent catalysts and the CNTs formed were studied using various characterization techniques. XRD and TPR analysis confirmed alloy formation in the bimetallic catalysts. The degrees of graphitization and crystallinity of CNTs formed over various catalysts were calculated using XRD and Raman analysis and were correlated with the catalyst properties. The structural morphology (fishbone or parallel wall type) and growth mechanism (tip or base growth) of the CNTs formed were investigated using TEM analysis. TGA analysis confirmed the absence of amorphous carbon formation during methane decomposition over Ni-, Fe- and Co-based catalysts. We also performed a few preliminary studies to selectively synthesise base grown CNTs over a Fe/SiO2 catalyst, which are considered to be easier to harvest, and also better for regeneration of catalyst compared to tip grown CNTs.


 Please wait while we load your content...
Please wait while we load your content...