Platinum-supporting hollandite-type vanadium–chromium mixed oxides as efficient heterogeneous catalysts for deoxygenation of sulfoxides under atmospheric H2 pressure†
Abstract
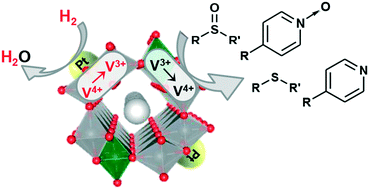
Deoxygenation of sulfoxides to their corresponding sulfides is an important reaction, and the development of efficient heterogeneous catalysts that can utilize molecular hydrogen (H2) as a reducing agent is highly desired. In this study, we successfully developed a new concept in heterogeneous catalysts, platinum-supporting hollandite-type vanadium–chromium mixed oxide catalysts (Pt/V(1−x)Crx-Hol, x: Cr/(V + Cr) molar ratio of the precursor solution), for sulfoxide deoxygenation with H2. Hollandite-type vanadium–chromium mixed oxides (V(1−x)Crx-Hol) that did not support platinum could stoichiometrically deoxygenate methyl phenyl sulfoxide to methyl phenyl sulfide, and the V3+ species in V(1−x)Crx-Hol were oxidized to V4+ after deoxygenation. However, the V4+ species could not be re-reduced with H2. By supporting platinum nanoparticles on V(1−x)Crx-Hol, the re-reduction of the oxidized vanadium species with H2 became possible. Thus, in the presence of Pt/V(1−x)Crx-Hol, catalytic sulfoxide deoxygenation with H2 as the reducing agent can be realized; the deoxygenation occurs on V(1−x)Crx-Hol, and the role of platinum is to re-reduce the oxidized vanadium species with H2. In the presence of Pt/V0.7Cr0.3-Hol, various types of structurally diverse sulfoxides could be selectively converted into their corresponding sulfides under atmospheric H2 pressure (1 atm). In addition, the present system was applicable to the deoxygenation of pyridine N-oxides to their corresponding pyridines. The observed catalysis was truly heterogeneous, and the Pt/V0.7Cr0.3-Hol catalyst could be reused for sulfoxide deoxygenation, although the performance was reduced.


 Please wait while we load your content...
Please wait while we load your content...