Yolk–shell-structured mesoporous silica: a bifunctional catalyst for nitroaldol–Michael one-pot cascade reaction†
Abstract
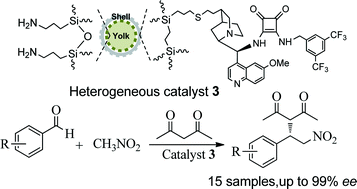
Great interest in heterogeneous asymmetric catalysis has focused on obtaining an enantioselective cascade reaction through a controllable active site-isolated heterogeneous catalyst. Herein, we utilize a yolk–shell-structured mesoporous silica and assemble an active site-isolated bifunctional heterogeneous catalyst, where chiral cinchonine-based squaramide molecules are anchored within a silicate channel as an outer shell while amine-functionalities are entrapped onto a silicate yolk as an inner core. Structural analyses and characterizations of the heterogeneous catalyst reveal its well-defined single-site chiral active species within its silicate network. Electron microscopy confirms the yolk–shell-structured mesoporous material. As presented in this study, as a bifunctional heterogeneous catalyst, it enables an efficiently nitroaldol–Michael cascade reaction to conduct the three-component coupling of nitromethane, aldehyde and acetylacetone into various chiral diones with high yields and up to 99% enantioselectivities in a one-pot process. As expected, this active site-isolated catalyst not only enhances the catalytic selectivity of the first-step nitroaldol condensation, but also keeps the enantioselectivity of the second-step Michael addition. Moreover, the heterogeneous catalyst can be also recovered easily and recycled repeatedly, making it an interesting feature in a three-component organic transformation.

 Please wait while we load your content...
Please wait while we load your content...