Pd–In intermetallic alloy nanoparticles: highly selective ethane dehydrogenation catalysts†
Abstract
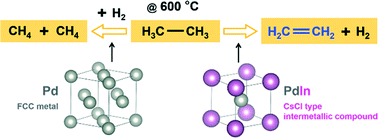
Silica supported Pd and Pd–In catalysts with different In : Pd atomic ratios and similar particle size (∼2 nm) were tested for ethane dehydrogenation at 600 °C. For a monometallic Pd catalyst, at 15% conversion, the dehydrogenation selectivity and initial turnover rate (TOR, per surface Pd site) were 53% and 0.03 s−1, respectively. Addition of In to Pd increased the dehydrogenation selectivity to near 100% and the initial TOR to 0.26 s−1. Carbon monoxide IR, in situ synchrotron XAS and XRD analysis showed that for Pd–In catalysts with increasing In loading, different bimetallic structures were formed: at low In loading a fraction of the nanoparticle surface was transformed into PdIn intermetallic compound (IMC, also known as intermetallic alloy) with a cubic CsCl structure; at higher In loading, a Pd-core/PdIn-shell structure was formed and at high In loading the nanoparticles were pure PdIn IMC. While a Pd metal surface binds CO predominantly in a bridge fashion, the PdIn IMC predominantly binds CO linearly. Formation of the PdIn IMC structure on the catalyst surface geometrically isolates the Pd catalytic sites by non-catalytic, metallic In neighbors, which is suggested to be responsible for the high olefin selectivity. Concomitant electronic effect due to Pd–In bond formation likely leads to the increase in TOR. Though multiple IMC structures with different atomic ratios are possible for the Pd–In binary system, only a cubic PdIn IMC with CsCl structure was observed, implying a kinetically controlled solid state IMC formation mechanism.
- This article is part of the themed collection: Nanocatalysis

 Please wait while we load your content...
Please wait while we load your content...