Pd/tetraphosphine catalytic system for Cu-free Sonogashira reaction “on water”†
Abstract
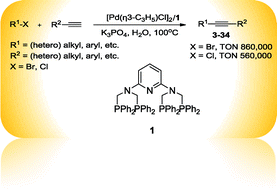
An efficient copper-free Sonogashira coupling reaction was performed on water at 100 °C with N,N,N′,N′-tetra(diphenylphosphinomethyl)pyridine-2,6-diamine (1) as a ligand, [Pd(η3-C3H5)Cl]2 as a catalyst precursor and K3PO4 as a base. Both aryl and heteroaryl halides were successfully alkynylated in this system, and a high turnover number (TON) up to 860 000 was obtained with a catalyst loading as low as 1 ppm.

 Please wait while we load your content...
Please wait while we load your content...