Proline hydroxylation at different sites in hypoxia-inducible factor 1α modulates its interactions with the von Hippel–Lindau tumor suppressor protein†
Abstract
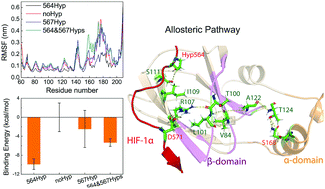
Hypoxia-inducible factor 1 (HIF-1) plays an essential role in the regulation of hypoxia in humans. This regulation is mediated by the interaction of the von Hippel–Lindau tumor suppressor protein (pVHL) with the hydroxylated HIF-1α at proline564 (Pro564). Experimental studies reported that Pro567 could also be hydroxylated. However, the conformational dynamics of the complex of pVHL with hydroxylated HIF-1α at Pro564 is not well understood, and whether hydroxylated Pro567 plays the similar essential role as Pro564 in regulating HIF-1α–pVHL interaction remains elusive. Herein, we performed all-atom molecular dynamics (MD) simulations on the pVHL/HIF-1α complexes with single hydroxylation at Pro564 and Pro567, double hydroxylation at both Pro564 and Pro567, and without hydroxylation. Our multiple MD simulations and binding energy calculations show that hydroxylation at Pro567 is less favorable for the binding of HIF-1α to pVHL, whereas hydroxylation at Pro564 results in an increase of structural rigidity of the pVHL/HIF-1α complex and an enhancement of the interactions between HIF-1α and pVHL. The different roles revealed here for Pro564 and Pro567 in regulating HIF-1α–pVHL interactions, together with the previous finding that HIF-prolyl hydroxylase PHD-3 participates in a negative feedback loop controlling the HIF-1 level, suggest that hydroxylated HIF-1α at Pro567 may perturb or may not participate in this negative feedback loop. Intriguingly, our simulation data and community network analysis demonstrate that the binding of hydroxylated HIF-1α at Pro564 to the β-domain of pVHL allosterically induces the conformational change of the α-domain via an optimal communication pathway from Pro564 of HIF-1α to S168 of the pVHL α-domain. This study reveals the different roles of Pro564 and Pro567 hydroxylation in HIF-1α in HIF-1α–pVHL interactions, which will be beneficial for developing effective strategies to treat hypoxia-related diseases and understanding the molecular basis of hypoxic training/exercise.


 Please wait while we load your content...
Please wait while we load your content...