Separation of water–ethanol solutions with carbon nanotubes and electric fields†
Abstract
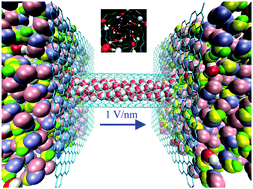
Bioethanol has been used as an alternative energy source for transportation vehicles to reduce the use of fossil fuels. The separation of water–ethanol solutions from fermentation processes is still an important issue in the production of anhydrous ethanol. Using molecular dynamics simulations, we investigate the effect of axial electric fields on the separation of water–ethanol solutions with carbon nanotubes (CNTs). In the absence of an electric field, CNT–ethanol van der Waals interactions allow ethanol to fill the CNTs in preference to water, i.e., a separation effect for ethanol. However, as the CNT diameter increases, this ethanol separation effect significantly decreases owing to a decrease in the strength of the van der Waals interactions. In contrast, under an electric field, the energy of the electrostatic interactions within the water molecule structure induces water molecules to fill the CNTs in preference to ethanol, i.e., a separation effect for water. More importantly, the electrostatic interactions are dependent on the water molecule structure in the CNT instead of the CNT diameter. As a result, the separation effect observed under an electric field does not diminish over a wide CNT diameter range. Moreover, CNTs and electric fields can be used to separate methanol–ethanol solutions too. Under an electric field, methanol preferentially fills CNTs over ethanol in a wide CNT diameter range.

 Please wait while we load your content...
Please wait while we load your content...