Identification of photofragmentation patterns in trihalide anions by global analysis of vibrational wavepacket dynamics in broadband transient absorption data†
Abstract
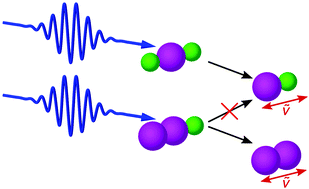
Trihalide anions are linear molecules that can be photodissociated with ultraviolet (UV) light. Whereas deep-UV excitation leads to three-body dissociation, for near-UV excitation just one molecular bond is cleaved, which notionally opens up the possibility for different fragmentation patterns. Here, we explore whether the dihalide fragment is formed as an anionic or neutral species and whether heteronuclear trihalides can lead to two different dihalides. The analysis is based on pronounced wavepacket dynamics induced by femtosecond UV pulses and associated both with the initial trihalide and the nascent dihalide species. For the trihalide anions I3−, Br3−, IBr2−, and ICl2− (point group D∞h), as well as for I2Br− and I2Cl− (point group C∞v) in dichloromethane solution, we identify dihalide fragments by their characteristic vibrational wavenumbers, which we achieve from globally fitting the vibrational wavepacket oscillations, considering a wavelength-dependent phase. No signature from neutral species is found right after excitation, hence there is only one diatomic product in D∞h trihalides. For the investigated C∞v trihalides, which could allow a homonuclear and a heteronuclear product, only the homonuclear one is observed. Since dihalide anions are unstable intermediates, their absorption and the ground-state bleach of the trihalide anion show a biexponential recovery for all samples due to recombining fragment pairs. The rate of the electron transfer yielding a neutral dihalogen and an atomic anion, a prerequisite for the recombination, gives rise to the biexponential behavior; fast recombination is mediated by vibrational excess energy, while slow recombination occurs for cooled-down dihalogens. These data reveal the fragmentation and recombination dynamics from a time-domain approach rather than frequency-domain vibrational spectroscopy and contribute to the in-depth comprehension of these versatile model molecules.

 Please wait while we load your content...
Please wait while we load your content...