New insights into the mixing of gold and copper in a nanoparticle from a structural study of Au–Cu nanoalloys synthesized via a wet chemistry method and pulsed laser deposition†
Abstract
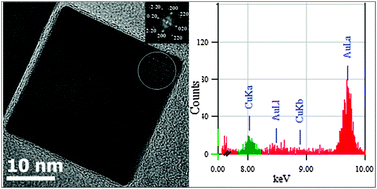
Gold–copper nanoparticles (Au–Cu NPs) were elaborated by both chemical (polyol reduction method) and physical (laser deposition) routes. The size, composition and crystal structure of these bimetallic nanoalloys were then characterized by aberration corrected transmission electron microscopy (TEM). Using a one-pot polyol method, Au–Cu nanocubes (NCs) with nominal compositions Au3Cu and AuCu3 were synthesized. The size and composition of the NCs were tuned by varying the amount and the ratio of Au(III) and Cu(II) ions used as metallic precursors in the reaction. While the particle shape and size were well-controlled, single particle X-ray spectroscopy showed that, irrespective of the targeted compositions, the Cu content in all NCs was about 11–12 at%, i.e. in both samples, the real composition was different from the nominal one. This was ascribed to an incomplete alloying of the two constituent metals of the alloy in the cubes due to different reduction kinetics of the two metallic precursors. To shed light on the alloying of gold and copper at the nanoscale, Au–Cu NPs with targeted compositions Au3Cu and AuCu3 were deposited on amorphous carbon by laser ablation of two monometallic sources, and their structural properties were studied by TEM. These studies show that Au–Cu nanoalloys were synthesized in both samples and that the complete mixing of Au and Cu atoms achieved with this synthesis technique led to the production of Au–Cu NPs with well-controlled compositions. These results constitute a first but major step towards a complete understanding of the details of kinetics and thermodynamics determining the mixing of gold and copper atoms at the nanoscale. Such an understanding is essential for producing Au–Cu bimetallic nanoalloys with well-defined structural properties via wet chemical synthesis.
- This article is part of the themed collection: Recent advances in the chemical physics of nanoalloys

 Please wait while we load your content...
Please wait while we load your content...