A study of the relationship between water and anions of the Hofmeister series using pressure perturbation calorimetry†
Abstract
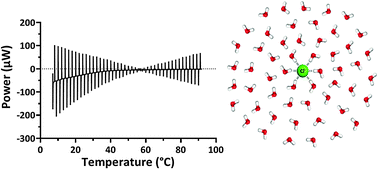
Pressure perturbation calorimetry (PPC) was used to study the relationship between water and sodium salts with a range of different anions. At temperatures around 25 °C the heat on pressurisation (ΔQ) from 1 to 5 bar was negative for all solutions relative to pure water. The raw data showed that as the temperature rose, the gradient was positive relative to pure water and the transition temperature where ΔQ was zero was related to anion surface charge density and was more pronounced for the low-charge density anions. A three component model was developed comprising bulk water, the hydration layer and the solute to calculate the molar expansivity of the hydration layer around the ions in solution. The calculated molar expansivities of water in the hydration layer around the ions were consistently less than pure water. ΔQ at different disodium hydrogen phosphate concentrations showed that the change in molar enthalpy relative to pure water was not linear even as it approached infinite dilution suggesting that while hydration layers can be allocated to the water around ions this does not rule out interactions between water and ions extending beyond the immediate hydration layer.

 Please wait while we load your content...
Please wait while we load your content...