Can metal-free silicon-doped hexagonal boron nitride nanosheets and nanotubes exhibit activity toward CO oxidation?†
Abstract
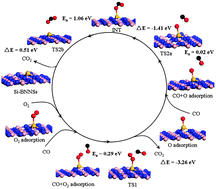
Si-doped hexagonal boron nitride nanosheets (Si-BNNS) and nanotubes (Si-BNNT) have been investigated by first-principle methods. The strong interaction between the silicon atom and the hexagonal boron nitride nanosheet or nanotube with a boron vacancy indicates that such nanocomposites should be very stable. The significant charge transfer from the Si–BNNS substrate to the O2 molecule, which could occupy the antibonding 2π* orbitals of O2, results in the activation of the adsorbed O2. The catalytic activity of the Si–BNNS for CO oxidation is explored and the calculated barrier (0.29 eV) of the reaction CO + O2 → CO2 + O is much lower than those on the traditional noble metals. This opens a new avenue to fabricate low cost and high activity boron nitride-based metal-free catalysts.

 Please wait while we load your content...
Please wait while we load your content...