Competition between n → πAr* and conventional hydrogen bonding (N–H⋯N) interactions: an ab initio study of the complexes of 7-azaindole and fluorosubstituted pyridines†
Abstract
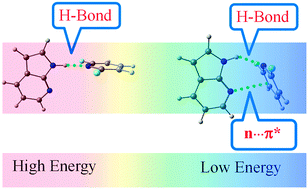
In this work, we have investigated a subtle competition between a very weak n → πAr* interaction and a very strong hydrogen bond (N–H⋯N) interaction present in the complexes of 7-azaindole with a series of 2,6-substituted fluoropyridines and observed how the weak interaction modulates the overall structural motif of these complexes in the presence of the strong interaction. We have studied the structures and binding energies of these complexes using MP2 as well as dispersion-corrected DFT calculations. It has been found that the strength of the N–H⋯N interaction in these complexes decreases with increasing fluorination in the fluoropyridine ring while the proximity between the nitrogen atom in 7-azaindole and the aromatic ring of fluoropyridine increases through n → πAr* interaction. Comparison of the binding energy values as well as structural parameters of these complexes calculated at the B3LYP level with those obtained at the MP2, M05-2X, and B97-D levels of theory clearly indicates that the dispersion effect is mostly responsible for this attractive n → πAr* interaction. This conclusion is also supported by localized molecular orbital-energy decomposition analysis (LMO-EDA). The current investigation is the first theoretical study on the n → πAr* interaction in the presence of a conventional strong hydrogen bonding interaction in the molecular system. Thus the present study has great significance for understanding the structures of the biomolecules as well as materials, as these interactions are very often present there simultaneously.

 Please wait while we load your content...
Please wait while we load your content...