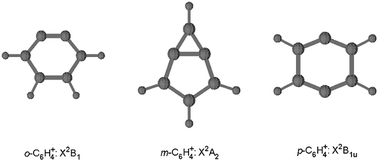
On the basis of the CASPT2 (multiconfigurational second-order perturbation theory) geometry optimization calculations, the ground states of the o-C6H4+ (C2v), m-C6H4+ (C2v), and p-C6H4+ (D2h) radical cations were determined to be 1 2B1, 1 2A2, and 1 2B1u, respectively. For o-C6H4+ and m-C6H4+, the first excited states (1 2A2 and 1 2A1, respectively) lie very close to the respective ground states. The small distance value of 1.419 Å between the two dehydrocarbons in the ground-state geometry of m-C6H4+ indicates that there is a real chemical bond between the two dehydrocarbons (the distance in the 1 2A1 geometry of m-C6H4+ is very long as in the m-C6H4 molecule). The (U)B3LYP isotropic proton hfcc (hyperfine coupling constant) calculation results imply that the ground and first excited states of o-C6H4+ will have similar ESR spectrum patterns while the ground and first excited states of m-C6H4+ will have completely different ESR spectrum patterns.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...