Morphology-selective synthesis of Cu(NO3)2·2.5H2O micro/nanostructures achieved by rational manipulation of nucleation pathways and their morphology-preserved conversion to CuO porous micro/nanostructures†
Abstract
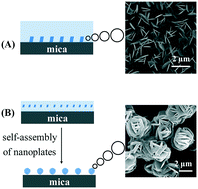
A special Cu(NO3)2 thin solution layer was built on highly hydrophilic mica surfaces and subjected to rapid evaporation. Controlling the evaporation time can intentionally manipulate the supersaturation. Morphology-selective synthesis of nanoplate- or microsphere-like Cu(NO3)2·2.5H2O micro/nanostructures has been succeeded by alternatively boosting either the heterogeneous nucleation at the substrate–solution interface at a low supersaturation level or the homogeneous nucleation in the bulk solution phase at a high supersaturation level. Our study presents a new method for morphology-selective synthesis of micro/nanostructures, which should also be applicable to materials other than Cu(NO3)2·2.5H2O and shall actually promote their applications. As a demonstration, Cu(NO3)2·2.5H2O micro/nanostructures were converted into porous CuO with well-preserved starting morphologies. The hierarchical porous CuO micro/nanostructures exhibited easily recyclable high photocatalytic activity towards visible-light degradation of methylene blue (MB).


 Please wait while we load your content...
Please wait while we load your content...