Construction and magnetic study of two new dysprosium complexes with chain or tetranuclear structure†
Abstract
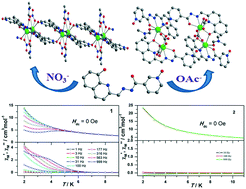
Two novel dysprosium complexes, namely, [Dy2L2(NO3)2(DMF)2]·2DMF (1) and [Dy4L4(OAc)4(DMF)2] (2), have been successfully synthesized by reacting a new pyridine-N-oxide-containing organic ligand N′-(8-hydroxyquinoline-2-carboxaldehyde)pyridine-N-oxide-carbohydrazide (H2L) with different dysprosium salts. The single-crystal X-ray diffraction study revealed that they show either one-dimensional (1D) or tetranuclear structure that is governed by the coordination anion used during the reaction. Within the 1D structure of 1, the nine-coordinated Dy3+ ions are bridged by two phenol oxygen atoms from the 8-hydroxyquinoline group to form Dy2 dimers and these dimers are further connected by pyridine-N-oxide groups. While in 2, the two octa-coordinated Dy3+ ions are bonded together by both phenol oxygen and acetate bridges to produce the Dy2 units and two adjacent Dy2 units are further linked by pyridine-N-oxide groups, resulting in a tetranuclear structure. Magnetic studies indicate that the anisotropic Dy3+ ions in 1 and 2 are ferromagnetically and anti-ferromagnetically coupled, respectively. The alternating current (ac) susceptibility measurements revealed slow magnetic relaxation with effective energy barriers of Ueff = 18.0 K under zero dc field in complex 1. However, under zero dc field, the ac data for 2 show negligible out-of-phase signals, excluding the existence of slow magnetic relaxation. Theoretical studies indicate that the different magnetic relaxation behaviors of both complexes were attributed to the different coordination environments around Dy3+ ions and the distinct magnetic coupling between them.


 Please wait while we load your content...
Please wait while we load your content...