Magnetism in CoFe2O4 nanoparticles produced at sub- and near-supercritical conditions of water†
Abstract
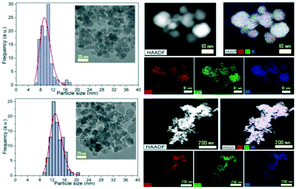
Formation of CoFe2O4 nanoparticles has been investigated via three hydrothermal synthesis routes with substantially different heating rates. The syntheses were conducted at various temperatures in the interval 100–390 °C using three different rector setups: 1) a conventional Teflon-lined steel autoclave, 2) a custom-designed spiral reactor and 3) a continuous flow reactor. A careful structural analysis was performed by powder X-ray diffraction and the crystallite size was determined by peak profile analysis employing both Rietveld refinement and whole powder pattern modelling. Transmission electron microscopy was used for morphology characterization and estimation of the particle size distribution. Complementarily, the crystallites' size distribution was determined by using the whole powder pattern modelling method. The crystallite sizes are highly dependent on the type of reactor used, and crystallites in the range of 8–15 nm with different morphologies were synthesized. The continuous flow reactor produced a narrow size distribution compared with the other two types of reactors. At room temperature, the as-prepared samples exhibit coercive field values from low (0.2 kOe) to moderate (1.2 kOe). The largest coercive value of 1230 Oe was obtained for the sample synthesized in an autoclave reactor at 240 °C for 1 hour. Zero-field-cooled/field-cooled susceptibility measurements revealed the blocking temperature to be above room temperature and a strong interparticle interaction was confirmed by Henkel plots. The Curie temperatures measured for the nanoparticles are either comparable or lower than that of the bulk, depending on the particle size.


 Please wait while we load your content...
Please wait while we load your content...