A new approach to facilely synthesize crystalline Co2(OH)3Cl microstructures in an eggshell reactor system†
Abstract
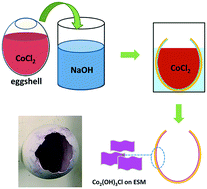
Sophisticated biological systems developed over millions of years of evolution can be rich sources of inspiration for engineers and scientists. There is increasing interest in the synthesis and applications of metallic hydroxy salts, for example Co2(OH)3Cl. It is not an easy task to prepare phase-pure structured Co2(OH)3Cl by direct mixing aqueous solutions of CoCl2 salts and NaOH in a flask. We demonstrate that crystalline Co2(OH)3Cl microparticles with unique morphology can be synthesized in eggshell reactor systems. The eggshell reactor system can provide the unique reaction environment for the formation of crystalline Co2(OH)3Cl microparticles with relatively well-defined shapes. Time-course experiments and intensive characterization helped us to propose a plausible formation mechanism. Our preliminary results suggest that the microscale Co2(OH)3Cl particles synthesized from the eggshell reactor system can be used for lithium-ion batteries, although its performance may still need to be further optimized and improved. In another attempt, we transformed the as-prepared Co2(OH)3Cl microparticle precursor into high-order structured Co3O4 microparticles preserving the same microscale morphology but with small Co3O4 nanoparticles as the basic building units. The as-prepared high-order structured Co3O4 microparticles demonstrate reasonably good electrochemical performance with a capacity of 900 mA h g−1 in reversible lithium ion storage.


 Please wait while we load your content...
Please wait while we load your content...