Dinuclear organogold(i) complexes bearing uracil moieties: chirality of Au(i)–Au(i) axis and self-assembly†
Abstract
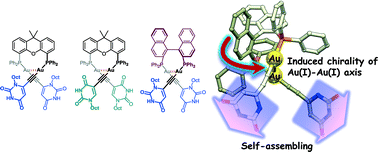
The conjugation of dinuclear organogold(I) complexes with a bridging diphosphine ligand as an organometallic compound and the uracil derivative as a nucleobase was demonstrated to afford bioorganometallic conjugates. Single-crystal X-ray structure determination of the dinuclear organogold(I)–uracil conjugates revealed the assembly properties of gold(I) and the uracil moieties in the solid state. The crystal structure of (U6Au)2(μ-Xantphos) (U6 = 6-ethynyl-1-octyluracil) with Xantphos as the bridging diphosphine ligand revealed the presence of an intramolecular aurophilic Au(I)–Au(I) interaction. R- and S-enantiomers based on the Au(I)–Au(I) axis exist in the unit cell, which are connected alternately to form the hydrogen-bonded assembly through intermolecular hydrogen bonds between the uracil moieties. In the case of the dinuclear organogold(I) complex (U5Au)2(μ-Xantphos) (U5 = 5-ethynyl-1-octyluracil), both enantiomers were found to form homochiral RR and SS dimers, respectively, through π–π interactions between 5-ethynyl-uracil moieties. In the crystal packing, each dimer is assembled alternately to form the hydrogen-bonded assembly through intermolecular hydrogen bonds between the uracil moieties. As expected, the utilization of (R)-BINAP as a bridging diphosphine ligand with axial chirality induced the chirality of the Au(I)–Au(I) axis. The crystal structure of the dinuclear organogold(I) complex with (R)-BINAP (U6Au)2(μ-R-BINAP) confirmed the axial chirality of the Au(I)–Au(I) axis, forming a R,R-enantiomer, wherein each molecule is arranged through intermolecular hydrogen bonds between the uracil moieties to form a helical molecular arrangement.

 Please wait while we load your content...
Please wait while we load your content...