Crystal nucleation of salicylic acid in organic solvents
Abstract
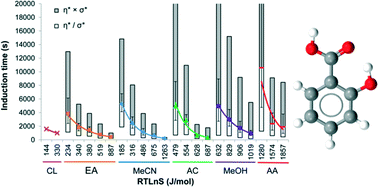
The crystal nucleation of salicylic acid was explored in a range of solvents using induction time and metastable zone width measurements. In total 3100 experiments were performed to collect statistically valid nucleation results. The lognormal cumulative probability function provided a representative fit for both induction time and metastable zone width distributions. At equal driving force the induction time is found to increase in the order chloroform, ethyl acetate, acetonitrile, acetone, methanol and acetic acid, and this order agrees with the order of increasing interfacial energy The metastable zone width (MSZW) value (expressed as supersaturation driving force) was highest in acetic acid followed by a lower value in methanol, consistent with the induction time results. In ethyl acetate, acetonitrile and acetone the corresponding MSZW values were lower but the order among these three solvents varied depending on the cooling rate and saturation temperature. A novel format for comparing the induction time and MSZW experiments is presented. The analysis reveals that the time of nucleation in the metastable zone width experiments is also dependent on the time of transforming clusters into nuclei, and not only governed by the rate of supersaturation generation. The relative influence of this transformation time depends on the solvent and the cooling rate.

 Please wait while we load your content...
Please wait while we load your content...