Porous coordination polymers based on azamacrocyclic complex: syntheses, solvent-induced reversible crystal-to-crystal transformation and gas sorption properties†
Abstract
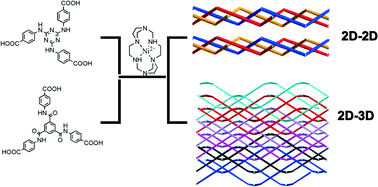
Three microporous coordination polymers, [NiL]3[(NiL)(H2O)2][(NiL)(TATAB)2]2·46H2O (1), [(NiL)3(TATAB)2]·5DMF (2) and [(NiL)3(BTCMT)2]·DMF·16H2O (3) (L = 1,3,6,9,11,14-hexaazatricyclo[12.2.1.16,9]octadecane, H3TATAB = 4,4′,4′′-triazine-1,3,5-triyltriaminobenzoic acid and H3BTCMT = 4,4′,4′′-[1,3,5-benzenetriyltris(carbonylimino)]-trisbenzoic acid), were constructed from two tripodal carboxylic ligands and an azamacrocyclic complex. The solvent-mediated, reversible, crystal-to-crystal transformation between 1 and 2 was achieved by immersing the crystalline samples in the corresponding solvent (H2O or DMF). Furthermore, during the fast bidirectional transformation, solvatochromic behavior was observed and confirmed. The comparison between 2 and 3 indicates that the degree of 63 monolayer corrugation has a significant effect on the formation and porous stability of an overall 2D or 3D network. Both 2 and 3 consist of large solvent accessible voids, but only compound 3 possesses permanent porosity, as confirmed by gas adsorption measurements and X-ray powder diffraction. In particular, compound 3 shows a high selective adsorption for CO2.

 Please wait while we load your content...
Please wait while we load your content...