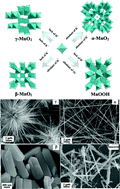
A hydrothermal process has been used to synthesize manganese oxides of various crystalline structures and morphologies, such as α-, β-, γ-MnO2, MnOOH and Mn3O4. The nanostructured materials were characterized by X-ray powder diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM), Raman spectroscopy, X-ray photoelectron spectroscopy (XPS) and hydrogen temperature-programmed reduction (H2-TPR) techniques. The crystalline evolution mechanism of converting β-MnO2 or MnOOH to α-MnO2 was studied. The crystalline-dependent effect of the nanostructured manganese oxides was explored by using toluene combustion as a probe reaction. Results indicated that the catalytic activity of α-MnO2 with ultra-long nanowires is higher than that of manganese oxides with other crystalline structures. The catalytic activity was correlated with the H2-TPR and XPS results.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...