Copper-catalyzed intermolecular asymmetric propargylic dearomatization of phenol derivatives†
Abstract
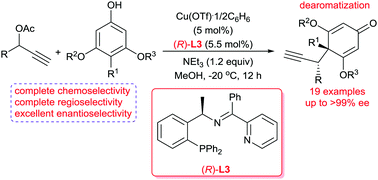
A copper-catalyzed intermolecular asymmetric propargylic dearomatization of phenol derivatives has been realized. Under the catalysis of Cu(OTf)·1/2C6H6 decorated with a chiral tridentate ketimine P,N,N-ligand, the dearomatization reaction proceeded smoothly with excellent control of chemo-, regio-, and enantioselectivities, thus providing a variety of optically active cyclohexadienone derivatives with up to >99% ee.


 Please wait while we load your content...
Please wait while we load your content...