Detection of small differences in the hydrophilicity of ions using the LCST-type phase transition of an ionic liquid–water mixture†
Abstract
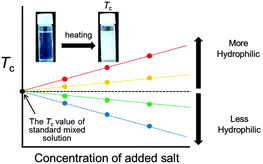
The phase separation temperature of the tetrabutylphosphonium trifluoroacetate–water mixture, which undergoes a lower critical solution temperature-type phase transition, is highly sensitive to the hydrophilicity of added salts; this system can be used to compare the hydrophilicity of the added ions.

 Please wait while we load your content...
Please wait while we load your content...